STANDARDIZATION OF A SPECTROPHOTOMETRIC PROCEDURE TO QUANTIFY THE ACTIVATION OF PNEUMOCOCCUS SEROTYPE 5 POLYSACCHARIDE
Keywords:
Streptococcus pneumoniae, Purpald assay, spectrophotometric, validation, aldehyde groups.Abstract
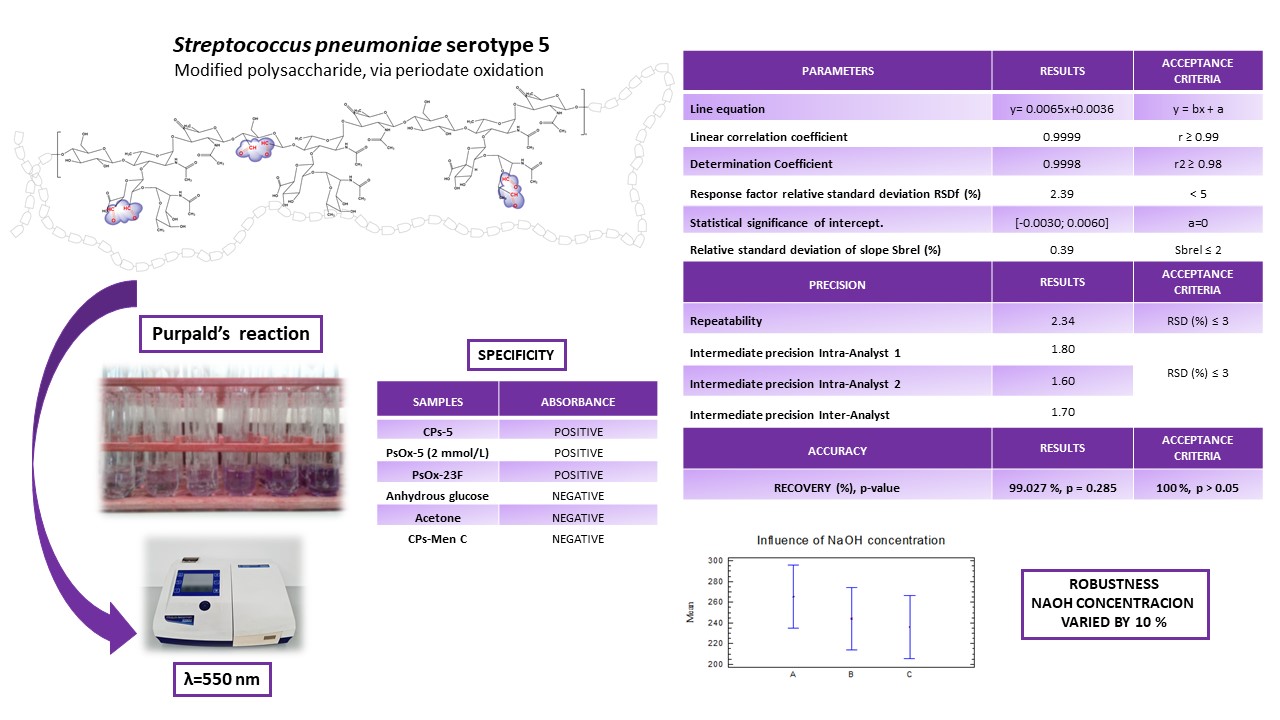
For the preparation of polysaccharide-protein conjugate vaccines, the hydrolysis and activation of capsular polysaccharides constitute a critical step for obtaining monovalent conjugates. This work describes the standardization of an analytical method for the quantification of aldehyde groups in the capsular polysaccharide of Streptococcus pneumoniae serotype 5, modified via periodate oxidation. The procedure is based on the reaction with Purpald's reagent. The proposed method was demonstrated to be linear (r² > 0,98, r > 0,99), with a fit coefficient of variation and a relative standard deviation of slope of less than 5 % and 2 %, respectively. It was precise, showing a relative standard deviation below 3 % under intermediate precision conditions, and robust against variations in NaOH concentration. The results in this study provide evidence for the safe use of the assay as process control in the modification step, of serotype 5 polysaccharide in the QuimiVio® vaccine.
References
LUNA, C. M. et al. “Decreased relative risk of pneumococcal pneumonia during the last decade, a nested case-control study”. Pneumonia, 2018, 10(1), 1-9. https://doi.org/10.1186/s41479-018-0053-6
CILLÓNIZ, C. et al. “Community-acquired pneumonia as an emergency condition”. Current opinion in critical care. 2018, 24(6), 531-539. DOI: https://doi.org/10.1097/MCC.0000000000000550
TILAHUN, M. et al. “High prevalence of asymptomatic nasopharyngeal carriage rate and Multidrug Resistance Pattern of Streptococcus pneumoniae among Pre-school Children in North Showa Ethiopia”. Infection and Drug Resistance. 2022, 15, 4253-4268. https://doi.org/10.2147/IDR.S377186
NAUCLER, P. et al. “Comparison of the Impact of Pneumococcal Conjugate Vaccine 10 or Pneumococcal Conjugate Vaccine 13 on Invasive Pneumococcal Disease in Equivalent Populations”. Clinical Infectious Diseases. 2017, 65(11), 1780-1790. https://doi.org/10.1093/cid/cix685
SANTIESTEBAN-LORES, L. E.; CABRERA-CRESPO, J.; CARVALHO, E. “Development of a pneumococcal conjugate vaccine based on chemical conjugation of polysaccharide serotype 6B to PspA”. Microbial Pathogenesis, 2021, 158, 105092. https://doi.org/10.1016/j.micpath.2021.105092
LISBOA, MP. et al. “Semisynthetic glycoconjugate vaccine candidate against Streptococcus pneumoniae serotype 5”. Proceedings of the National Academy of Sciences, 2017, 114(42), 11063-11068. https://doi.org/10.1073/pnas.1706875114
LEE, C. H.; TSAI, CH. M. “Quantification of bacterial lipopolysaccharides by the purpald assay: measuring formaldehyde generated from 2-keto-3-deoxyoctonate and heptose at the inner core by periodate oxidation”. Analytical biochemistry, 1999, 267(1), 161-168. https://doi.org/10.1006/abio.1998.2961
PORRO, M. et al. “Modifications of the Park-Johnson ferricyanide submicromethod for the assay of reducing groups in carbohydrates”. Analytical Biochemistry, 1981, 118(2), 301-306. https://doi.org/10.1016/0003-2697(81)90586-8
OLIVEIRA, F. S. et al. “Determination of total aldehydes in fuel ethanol by MBTH method: sequential injection analysis”. Journal of the Brazilian Chemical Society, 2005, 16, 87-92. https://doi.org/10.1590/S0103-50532005000100013
HLADOVÁ, M. et al. “Review of spectrophotometric methods for determination of formaldehyde”. Vedecké Práce Materiálovotechnologickej Fakulty Slovenskej Technickej Univerzity v Bratislave so Sídlom v Trnave, 2019, 27(44), 105-120. https://doi.org/10.2478/rput-2019-0012
WOOD-DUQUE, M. et al. “Validación del método de Ellman para la determinación de la concentración de grupos sulfhidrilos a muestras de la producción de la vacuna sintética contra el Haemophilus influenzae tipo b”. VacciMonitor, 2014, 23(2), 73-80. ISSN 1025-028X
CENTRO PARA EL CONTROL ESTATAL DE MEDICAMENTOS, EQUIPOS Y DISPOSITIVOS MÉDICOS (CECMED). Resolución CECMED No. 40/2014: Anexo No.1 de las Buenas Prácticas para Laboratorios de Control de Medicamentos, Validación de Métodos Analíticos. La Habana: CECMED; 2014. Available in: https://www.cecmed.cu/reglamentacion/aprobadas/resolucion-cecmed-40 (Consulted online: april 21, 2025).
WORLD HEALTH ORGANIZATION. The International Pharmacopoeia 11th edition. Geneva: WHO; 2022. ISBN 978-92-4-006243-6. Available in: https://digicollections.net/phint/. (Consulted online: december 17, 2025).
ABREU-VÁZQUEZ, J. C.; PÉREZ-HERRERA, J. Z.; MERCHÁN-MILIÁ, A. Y. “Validación del método colorimétrico de bifenilo para la determinación del contenido de carbohidratos en el polisacárido capsular de Streptococcus pneumoniae serotipo 5”. VacciMonitor, 2025, 34. ISSN 1025-0298.
MÁ PENSAMIENTO, S. F. “Validación de los métodos analíticos aplicados al agua potable utilizada en la elaboración de soluciones orales hidratantes de acuerdo a la Farmacopea USP XXV como método documentado de control para el mantenimiento de la calidad de la misma en Laboratorios Alfa Farmacéutica SA”. Tesis Doctoral. Universidad de San Carlos de Guatemala, Guatemala, 2015. Available in: https://core.ac.uk/download/pdf/35293068.pdf. (Consulted online: september 1, 2022).
LEE, C. H.; FRASCH, C. E. “Quantification of bacterial polysaccharides by the purpald assay: measurement of periodate-generated formaldehyde from glycol in the repeating unit”. Analytical Biochemistry, 2001, 296(1), 73-82. https://doi.org/10.1006/abio.2001.5230
KANDPAL, B. M. et al. “4-Amino-3-hydrazino-5-mercapto-1, 2, 4,-triazole (PURPALD) in organic chemicals analysis: Distinguishing aldehydes and ketones”. Journal of Integrated Science and Technology, 2024, 12(1), 709-709. ISSN: 2321-4635.
GARCÍA MARTÍNEZ, E. M. “Aplicación de la ley de Lambert-Beer en espectroscopía UV-visible”. 2012. Available in: https://riunet.upv.es/handle/10251/16360. Consulted online: september 1, 2022).
DALAL, J. et al. “Development and pre-clinical evaluation of a synthetic oligosaccharide-protein conjugate vaccine against Neisseria meningitidis serogroup C”. Vaccine, 2019, 37(36), 5297-5306. https://doi.org/10.1016/j.vaccine.2019.07.053
ZAKRZEWSKI, R; SKOWRON, M. “Application of Purpald" for determination of 3-cyclohexene-1carboxaldehyde and hydroxyisohexyl-3-cyclohexene carboxaldehyde”. J. Cosmet Sci, 2013, 64(5), 391-400. URL: https://library.scconline.org/v064n05/75
ZUREK, G.; KARST, U. “Microplate photometric determination of aldehydes in disinfectant solutions”. Analytica chimica acta, 1997, 351 (1-3), 247-257. https://doi.org/10.1016/S0003-2670(97)00363-2
BOTELHO, J. R. S. et al. “Development and validation of a UV-Vis spectrophotometric method for estimation of total content of chalcone”. Methods X, 2025, 14, 103119. https://doi.org/10.1016/j.mex.2024.103119
MIEDVIEDIEVA, K. P.; VASYUK, S. O.; PORTNA, O. O. “Development and validation of a new spectrophotometric method for the determination of gabapentin in capsules”. ScienceRise: Pharmaceutical Science. 2023, 43(3), 50-57. DOI: https://doi.org/10.15587/2519-4852.2023.283270
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Bárbara Baró-Bicet, Felix Cardoso-San Jorge, Raine Garrido-Arteaga, Darielys Santana-Mederos, Jessy Pedroso-Fernández

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This journal provides immediate open access to its content, based on the principle that offering the public free access to research helps a greater global exchange of knowledge. Each author is responsible for the content of each of their articles.






















